|

17K013,15%, 2 mEq/ml, 20 ml/Amp現行藥物外觀
中文名:氯化鉀 注射液
廠牌:信東
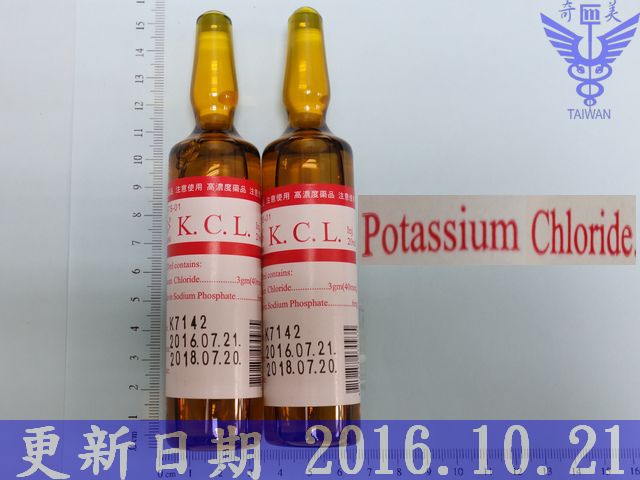
17K013,15%, 2 mEq/ml, 20 ml/Amp
中文名:氯化鉀 注射液
廠牌:
|
| Product
name |
KCL |
|
| Dosage
form |
| 17K013,
15%, 2 mEq/ml, 20 ml/Amp |
|
| Indications |
|
| Administration
& Dosage |
|
Adult
Hypokalemia:
Serum K< 2 mEq/L: IV, 20-40 mEq/hr, with continuous cardiac monitoring; Max. 400 mEq/d
Serum K> 2.5 mEq/L: IV, 10-15 mEq/hr; Max. 200 mEq/d
Pediatric
Hypokalemia: doses should be based on serum K levels
IV, 0.5-0.75 mEq/kg/dose; infused 0.3-0.5 mEq/kg/hr,
Max. 1 mEq/kg/hr and 30 mEq/dose; 3 mEq/kg/d or
40 mEq/m2/d. |
|
| Contraindication |
hyperkalemia; severe renal impairment;oliguria or azotemia;anuria;crush syndrome;untreated Addison's diseasea
adynamia episodica hereditoria;acute dehydration;heat cramps. |
|
| Precaution |
|
| Adverse
reaction |
| Diarrhea; flatulence; N/V; abdominal pain; cardiac arrest; ECG changes; GI ulceration; hyperkalemia |
|
| Drug
Interaction |
|
| Pregnancy
Category |
|
| Notes |
| Must dilute IV solution prior to use. Max. IV concentration = 40 mEq/L for peripheral IV line, to 80 mEq/L (with monitoring) through central line. Max. administration rate = 40 mEq/hr in adults |
|

